TREATMENT OPTIONS
The following agents are being evaluated in children with OMS for control of neurological symptoms. Some of them originally were used to treat cancer or organ transplant rejection, but their use has been broadened to difficult autoimmune disorders. In an effort to match the immunotherapy to your child's needs, different treatments are typically combined. For several, there are no published studies yet in OMS, but the approaches are novel and promising. You should fully explore the risks before deciding to participate in clinical trials.
For further information, click on the hyperlink for the specific agent.
Biologicals
Drugs
Other treatments
Devices for IV access
ACTH (Corticotropin) Treatment
ACTH is a hormone. Normally secreted by the pituitary gland, it stimulates the adrenal gland to make cortisol. ACTH is given by intramuscular injection for several weeks. A beneficial response usually occurs within the first two weeks of use. If there is no improvement during that time, contact your health care provider.
ACTHAR® gel. ACTH is currently available in gel form at 80 I.U. (international units) per cc. Due to short supply, it must be obtained from the National Organization for Rare Disease (NORD) by your physician.
ACTH dose. We advocate the use of a high dose protocol with slow tapering. With our protocol, the chances of inducing a remission are excellent. Other agents are usually necessary to maintain the remission during weaning from ACTH.
Possible initial side effects: ACTH may cause irritability, elevated blood pressure, salt and water retention, potassium and calcium loss, nausea, vomiting, or dizziness. These are reversible when the dose is lowered.
Possible side effects with long-term use: Because of possible long-term side effects, we do not recommend that ACTH be used daily beyond two weeks. Alternate day dosing helps prevent cataracts, glaucoma, increased risk for infection, osteopenia or osteoporosis, growth suppression, muscle weakness, metabolic problems, peptic ulcer, skin problems such as poor wound healing, facial redness, acne, fragile skin.
Baseline tests: Prior to starting ACTH, obtain blood and urine for a CBC and Chem 20 and a urinalysis.
Begin ACTH injections in the hospital. Our published protocol provides details of dosing. The nurse will give the first one and train you to give the second one. A blood pressure measurement is advisable before and an hour after each injection.
During the first month on ACTH your child will need:
1. Weekly urine checks for glucose
2. Weekly blood pressure checks
3. Weekly chemistry 20 blood draws
4. Weekly weights
Special information:
1. No immunizations should be given while child is on ACTH.
2. ACTH therapy may mask signs of infection. Be alert to symptoms of illness.
3. Salt and salty food causes fluid retention. Avoid them.
4. Call the physician if fever, infection, fluid retention, weight gain, muscle weakness, stomach/abdominal pain, seizures or headache occurs.
5. Your health care provider may want to start your child on an antacid to protect the stomach while the child is on ACTH.
6. Some insurances do not cover ACTH. NORD offers an ACTH financial assistance program for children who need it but are not covered by insurance.Advice for Long-Term Users:
1. Annual bone density test for osteoporosis
2. Annual eye examination
3. EKG or echocardiogram if tricyclic antidepressants or methylphenidate (Ritalin® ) are used
4. When ACTH is discontinued, take physiological steroid replacement for 6-12 months or as recommended by an endocrinologist
5. Any fracture should signal the possibility of osteoporosis
6. Take a daily calcium supplement.
Intravenous Immunoglobulin Therapy (IVIG)
Intravenous immunoglobulin (IVIG) is a preparation of immunoglobulins (antibodies) which are pooled from a variety of healthy human donors.
Different brands of IVIG are available. They differ in how they are prepared and in many other technical ways. When a child does not respond to IVIG or has side effects, a different brand should be tried.
Cytogam
Gamimune N S/D 10%
Gammagard S/D
Gammar-P I.V.
IVEEGAM EN
Pangloulin
Polygam S/D
Sandoglobulin
Venoglobulin-S (5% or 10%)
WinRho SD
For further information about IVIG, you may wish to contact:
Blood Diagnostics, Inc.
1-800-948-9834
www.ivig.com
The advantage of IVIG is that it has the potential to reduce or eliminate the need for drugs that have serious side effects. It is the only treatment for autoimmune disease that doesn't suppress the immune system and can be used in children whose immune systems are already suppressed.
IVIG can be combined with other therapies, such as ACTH, steroids, chemotherapy, and other immunosuppressants as well as plasmapheresis.
How is IVIG administered? As IVIG is a blood product, a needle must be inserted into the child's vein and the solution of immunoglobulins is dripped in slowly over a few to several hours. The usual dose is 1-2 g/kg.
How does IVIG work? IVIG probably has several mechanisms of action. It binds to autoantibodies and target cells and decreases levels of circulating immune complexes. It prevents the immune system from carrying out its attack.
Are there risks associated with IVIG? During or after an IVIG infusion, there can be nausea, vomiting, flu-like symptoms, fever, and headache, which are resolved by decreasing the infusion rate. If these symptoms occur 1-2 days after the infusion, they generally resolve within 24-48 hours with Tylenol. More serious complications are rare.
Where is IVIG given? The initial dose (if a child has never been on IVIG) should be administered in a hospital or outpatient clinic setting. Other treatments can be delivered at home by a registered nurse who is familiar with the protocol, prepared for adverse reactions, and available to the parent for support during and after the IVIG infusion.
How many IVIG treatments are required? The child may not show any response until 10-14 days from the first infusion. If there is a good response, IVIG should be given monthly. If there is no improvement, a higher dose should be used the following month. If there is no significant improvement after the second infusion, then IVIG should be discontinued.
What blood work is necessary for IVIG? Your local physician will need to draw some preliminary blood test and obtain a urine sample prior to starting the IVIG protocol. This is to check for kidney function and IgA, an immunoglobulin in the blood. If this blood work returns normal, then the child is entered into the IVIG protocol. A one-time urine sample will be needed after the treatment to monitor kidney function.
Rituxan is an IgG antibody against B cells. First used in 1987 to treat patients with B-cell lymphoma, it is now being studied in patients with autoimmune neurological disorders. Rituxan binds to the CD20 antigen on the surface of mature B cells, which it targets for immune-mediated destruction.
How is Rituxan given? 
Rituxan is given by intravenous infusion once a week for four doses. The dose is usually 375 (10-500) mg per square meter of body surface area. The initial rate of infusion should be 50 mg/hour. If there are no hypersensitivity reactions, the infusion can be increased stepwise by 50 mg/hour up to 400 mg/hr. However, if a hypersensitivity reaction occurs, the infusion should be slowed or stopped.
How long does the effect last?
Rituxan depletes circulating B cells and the effect can last 6-9 months. It also binds to lymphoid cells in the spleen, thymus, and lymph nodes. B cell recovery starts about 6 months after the completion of treatment and continues up to 12 months. Some patients show a reduction in levels of serum immunoglobulins (IgG and IgM) after treatment, but most do not.
What are the risks?
Rituxan is genetically engineered from murine cells, the proteins of which can cause hypersensitivity.
Within 30 minutes to 2 hours after the first infusion, most patients get fever and chills. Other infusion-related symptoms include nausea, hives, fatigue, headache, and itching. Symptoms resolve with slowing of the infusion and treatment with acetaminophen (Tylenol) and IV saline and Benadryl. Once patients have had this reaction, they are best pretreated for subsequent infusions.
Rituxan can be associated with more serious hypersensitivity reactions such as low blood pressure (hypotension), breathing difficulties (bronchospasm), and sensation of tongue or throat swelling (angioedema). Epinephrine, antihistamine, and corticosteroids should be on hand for treatment of hypersensitivity.
There is a small risk of bacterial blood infections (sepsis) and viral infections (herpes). All fevers or apparent infections should be reported to your physician.
How are drug effects monitored?
Complete blood counts (CBC) and platelet counts should be monitored at intervals. The monitoring of immunoglobulins and B cells is desirable.
Imuran is an extremely effective drug for treatment of certain immune diseases such as myasthenia gravis and polymyositis. Properly used, it is a very safe drug. However, its use requires cooperation of the patient, the physician, and the laboratory. The more you know about Imuran, the better you will be able to assure the success of your treatment.
What does Imuran do?
Imuran suppresses the immune system and the blood cells. In order to assure a proper degree of treatment, your child's blood must be checked periodically. These checks are done frequently at first, but can be carried out less frequently later on, after the dose of Imuran is established.
Starting your treatment with Imuran
Since some patients may be unusually sensitive to Imuran, we start treatment with a very low dose. You will be asked to report immediately if you experience any of the following possible side effects of the medication:
1. Fever
2. Rash
3. A flu-like feeling
4. Nausea, or abdominal discomfort
Adjusting your dose
This requires frequent checks of your blood. The dose of Imuran should be adjusted individually for every patient. Until the optimal dose is established, the blood test must be carried out at fairly frequent intervals (usually 1 to 2 weeks). The frequency of testing must be arranged between the patient and the physician.
Possible side-effects of the medication
Imuran may have the following side-effects:
1. Nausea or abdominal discomfort
2. Easy bruising or bleeding from the gums
3. Sore tongue or mouth
4. Other abnormalities that can only be detected by laboratory testing
Imuran can be used with your cooperation. Be sure that you understand how frequently you must be seen and how frequently your blood must be tested. Please be sure that laboratory reports get to your physician. If you have any questions about other illnesses, surgery, or the use of other drugs, call your physician.
What is Cytoxan (CTX)?
Cytoxan is an anticancer drug sometimes also used to treat certain autoimmune disorders that have not responded to treatment. It may be taken alone or in combination with other anticancer drugs. Cytoxan is one chemotherapeutic agent commonly used in the treatment of neuroblastoma.
How should the medication be taken?
Cytoxan comes in tablet form and an injectable form that can be made into an oral solution for children who can't swallow pills. The oral solution needs to be refrigerated and used within a two weeks. The usual pediatric dosage of Cytoxan is 1-5 mg per kg (2.2 pounds) of body weight. It is taken over a period of 60 to 90 days.
While taking Cytoxan, children must drink a 2 to 3 liters of fluid a day to help prevent injury to the bladder and eliminate Cytoxan's toxic byproducts. If you miss a dose, do not take 2 doses at once. Take Cytoxan on an empty stomach unless it causes severe stomach upset.
Will laboratory testing be required?
Frequent blood tests are needed to monitor the white blood cell count. If the number of white blood cells drops below a certain level, a reduction in dosage will be required. Frequent urine tests also check for blood in the urine, which would be a sign of bladder injury.
Who should not take this medicine?
Children with kidney or liver disease, previous anticancer chemotherapy or radiation therapy, blood disease with low white blood cell or platelet count, or bone marrow tumors are at increased risk for toxic side effects of Cytoxan.
Do not give your child Cytoxan if there has been an allergic reaction to it or other anticancer drugs, or if your child's bone marrow function is abnormal.
What are common side effects?
Loss of appetite, nausea, vomiting, constipation, and temporary hair thinning or brittleness are common side effects. Cytoxan can make your child more susceptible to infections by reducing the activity of the immune system.
Are there any important uncommon side effects?
Cytoxan may cause another type of cancer to form in the bladder, lymph nodes, or bone marrow even several years after the drug is given. It can be associated with abdominal pain, anemia, diarrhea, fever, mouth sores, poor wound healing, rash, severe allergic reaction, skin and fingernail darkening, and yellow jaundice.
What about drug interactions?
The dose of Cytoxan may need adjustment in children who also are taking Adriamycin, Allopurinol, or Phenobarbital.
Children on adrenal steroids who have had an adrenal gland removal are at increased risk for toxic side effects from Cytoxan. The dosages of both drugs may need to be altered.
No vaccinations should be given during treatment with Cytoxan.
When should I call my doctor?
If your child experiences any of the following symptoms, contact your health care provider immediately: chills, cough, black stools, dizziness, fever, rash, reddish urine, shortness of breath, side or back pain, swollen feet, unusual bruising or bleeding.
6-Mercaptopurine
(Puri-Nethol®)
Mercaptopurine is chemotherapy that is given as a treatment for some types of cancer and is also used as a "steroid sparer" to treat certain autoimmune diseases. This fact sheet describes mercaptopurine, how it is given and some of its specific side effects.
Each person's reaction to chemotherapy is unique. Some people have very
few side effects, while others may experience more. The side effects described
in this fact sheet will not affect everyone who is given mercaptopurine,
and may be different if you are having more than one chemotherapy drug.
We have outlined the most common and the less common side effects, so
that you can be aware of them if they occur. However, we have not included
those that are very rare and therefore extremely unlikely to affect you.
If you do notice any effects which you think may be due to the drug, but
which are not listed in the fact sheet, please discuss these with your
doctor or chemotherapy nurse.
You will see your doctor regularly while you have this treatment so that
they can monitor the effects of the chemotherapy. This fact sheet should
help you to discuss any queries about your treatment and its side effects
with your doctor or chemotherapy nurse, as they are in the best position
to help and advise you.
What it looks like
Mercaptopurine is available as beige colored tablets of 50mg.
How it is given
The tablets should be swallowed whole with plenty of water.
Possible side effects
Temporary reduction in the production of blood cells by the bone marrow.
This can result in anemia, risk of bruising or bleeding and infection.
The numbers of blood cells in your blood may begin to reduce from about
seven days after the treatment has been given and usually reaches its
lowest point at 10-14 days after the chemotherapy. The numbers of blood
cells will then increase steadily and will usually return to normal within
21-28 days.
The extent to which your blood cells are reduced depends on the dose of
chemotherapy you have and which other chemotherapy drugs, if any, are
given in combination. Your doctor can advise you how likely it is that
your blood cells will be lowered by the chemotherapy. Your blood will
be checked regularly to see how well your bone marrow is working.
If your temperature goes above 38C (100.5F), or you develop any unexplained
bruising or bleeding, or you suddenly feel unwell, even with a normal
temperature, contact your doctor or the hospital straight away.
Tiredness and a general feeling of weakness. It is important to
allow yourself plenty of time to rest.
Your liver may be temporarily affected. Mercaptopurine may cause
changes in the way that your liver works, which return to normal when
the treatment is finished. This is very unlikely to cause you any harm,
but your doctor will monitor this carefully. Samples of your blood will
be taken from time to time to check your liver function.
Less common side effects
Nausea (feeling sick) and vomiting. This is usually mild. There
are now very effective anti-sickness drugs to prevent or greatly reduce
this. If you do feel sick it may start from the time that the treatment
is given. If it is not controlled, or continues, tell your doctor. They
can prescribe other drugs, which may be more effective.
Sore mouth and taste change. Your mouth may become sore, or you
may notice small ulcers during this treatment. Drinking plenty of fluids
and cleaning your teeth regularly and gently with a soft toothbrush can
help to reduce the risk of this happening. Tell your doctor if you do
have any of these problems as they can prescribe special mouthwashes and
medicine to prevent or clear any mouth infection.
You may notice that your food tastes different. Normal taste will come
back after the treatment finishes.
Diarrhea. This can usually be easily controlled with medicine but
let your doctor know if it is severe or continues. It is important to
drink plenty of fluids if you do have diarrhea
Skin changes. Mercaptopurine can cause a rash, which may be itchy.
Your doctor can prescribe medicine to help with this.
Raised levels of uric acid in the blood. Mercaptopurine can cause
an increased level of uric acid in your blood. If so, the dose of mercaptopurine
must be adjusted accordingly. It also helps to drink plenty of fluids
to flush the uric acid through your body.
A high temperature and chills may occur, but they do not usually last
long. Your doctor may prescribe medicines to reduce these side effects.
Additional information
Some other medicines can be harmful to take when you are having chemotherapy.
Always let your doctor know of any other medicine you are taking.
Fertility. Your ability to become pregnant or father a child may
be affected by taking this drug. It is important to discuss fertility
with your doctor before starting treatment.
Contraception. It is not advisable to become pregnant or father
a child while taking mercaptopurine as it may harm the developing fetus.
It is important to use effective contraception while taking this drug,
and for at least a few months afterwards. Again, discuss this with your
doctor.
Things to remember about mercaptopurine tablets
- The tablets should be stored in a dry place at room temperature away from direct sunlight.
- It is important to take your tablets at the right times. You must take them as directed by your doctor.
- Keep the tablets in a safe place where children cannot reach them.
- If you are sick just after taking the tablet inform the doctor immediately, as you may need to take another dose. Do not take another tablet without first telling your doctor.
- If you forget to take a tablet do not take a double dose. Tell your
doctor and keep to your regular dose schedule
What is Apheresis?
The literal meaning of apheresis is "to take away." The term refers to extracorporeal immunotherapy, or the removal of blood components by passing the patient's blood through an external device. "Continuous" apheresis machines withdraw blood while also giving it back.
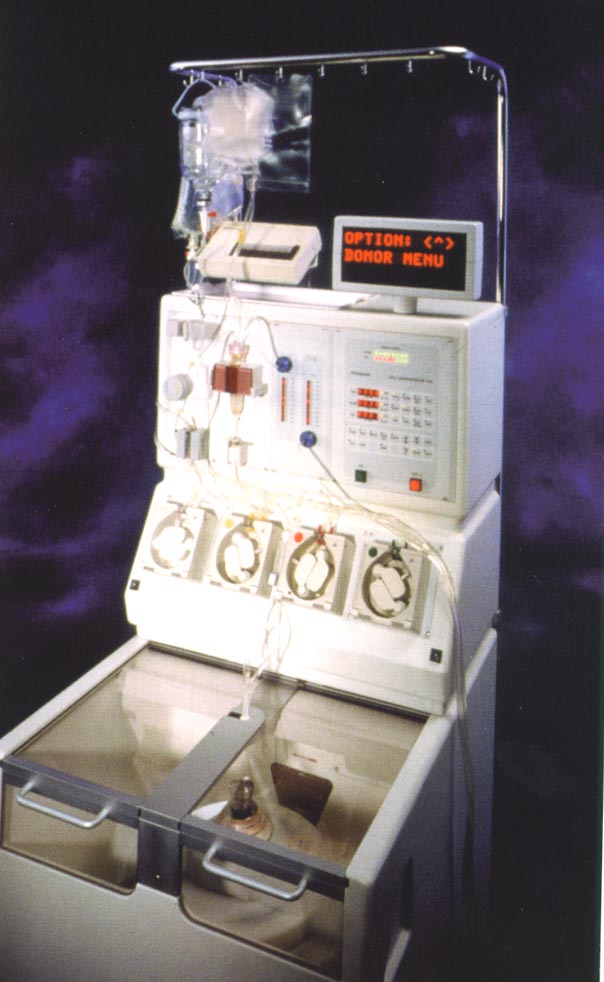
Is Apheresis Feasible in Children with Opsoclonus-Myoclonus?
The amount of blood that must be removed to "prime" the equipment (tubing, filters, columns, pump) is the limiting factor on how young a patient can be to receive apheresis. Children with opsoclonus-myoclonus, which usually appears between 18 to 24 months of age, are not good candidates for plasmapheresis because a minimum of 250-300 cc of blood must be removed, which is up to one-third of their blood volume.
Even if the extracorporeal circuit volume is > 10% of the patient's blood volume, as is the case for children under 20 to 25 kg, a transfusion with 1 unit of red blood cells will be necessary. Fluid shifts, reduced platelets, and anticoagulation are potential serious side effects. Although children as small as 10 kg have been treated by plasmapheresis, the procedure becomes extremely high risk and potentially lethal. Children weighing less than 40-50 kg probably require a central venous catheter.
What are the Main Types of Apheresis?
The three types of apheresis most relevant to opsoclonus-myoclonus are:
- Plasmapheresis
- Leukopheresis
- Immunoadsorption
What is Plasmapheresis (PP)?
For more than 30 years, plasmapheresis has been used to remove autoantibodies in various autoimmune diseases. Different methods have been applied, such as plasma exchange, double filtration, and immunoadsorption.
The amount of the apheresis is usually 1 plasma volume per session, and a minimum of 5 sessions is required. To replace the plasma removed, the patient receives replacement fluids, such as crystalloids and anticoagulant, and 5% albumin or fresh frozen plasma.
The most common side effect of plasmapheresis is low blood pressure (hypotension). It may be due to reduced blood volume (hypovolemia), the vasovagal reflex (like fainting), or activation of complement (an immune system component).
What is Leukopheresis (LP)?
The removal of white blood cells from the patient's blood is done by leukopheresis. One-half of patients have transient side effects, such as nausea, vomiting, chills, fever, respiratory distress, palpitations, chest oppression, and nasal stuffiness. Line clotting may be more common when heparin rather than another anticoagulant is used. Leukopheresis is usually used to lower grossly elevated white blood cell counts as in leukemia.
What is Immunoadsorption (IA)?
Antibodies can be removed from blood by immunoadsorption at specially trained and equipped apheresis centers. The procedure, which has been used for over 10 years, is the same as plasmapheresis except that the patient's blood is run through a column made of staphylococcal antigen (protein A). It's called an off-line procedure. Two types of columns are commercially available in the U.S.: Prosorba® and Immunosorba®.

Up to 90 % of immune complexes and IgG antibodies in the blood adhere to the column antigens, although not all types of IgG are as susceptible. The blood is then returned to the patient. In contrast to older columns, these columns contain highly purified protein A covalently attached to a silicon matrix, which means less leaching of the staphylococcal protein into the blood and no toxicity to the heart or lungs.
Immunoadsorption is often linked with cyclophosphamide (Cytoxan®) and steroid therapy. It has been used in many different neurological disorders and as a treatment for some tumors. Immunoadsorption modulates the humoral (antibodies) as well as cellular (T cells) immune system.
Using an adolescent as an example, one-half liter (500 ml) of blood is removed and centrifuged to separate the plasma from the cellular portion, which is then returned to the patient. The patient's plasma is perfused through the column and then reinfused into the patient at 10-20 ml/min. Because only a small portion of blood is treated at any session, multiple treatments are necessary. Six treatments (twice weekly for 3 weeks) are usually used.
Immunoadsorption columns cost $1,000 to $6,000 per column. The more expensive one is reusable for the same patient. Some insurance companies do cover the cost.
Catheters Used for IVIG or Cancer Treatment
Children who require repeated administration of IV agents may need to have a catheter placed in the chest or arm. The secured IV access allows IVIG, cancer chemotherapy, and parenteral nutrition to be infused.
What are the types of catheters?
- Internal
- External
What are internal catheters?
The most commonly used internal catheter is the Port-a-Cath, a subcutaneous port. It is hidden beneath the surface of the skin. There's no outward sign except a slight bump under the skin.
What are external catheters?
The most commonly used external catheters are the Broviac or Hickman catheters. Peripherally inserted central catheters are called PICC lines.
What are the pros and cons of these catheters?
Internal catheters are advantageous for children because they do not like to see tubes sticking outside of their bodies. However, kids with a tremendous fear of needles might not be good candidates because accessing the port requires a needle stick.
External catheters reduce the number of painful procedures. However, they require higher maintenance, daily care, and they cost more. Small children may dislodge or damage external catheters.
Do catheters cause complications?
Catheter complications vary with the type of catheter. There have been several large studies. With careful handling and proper selection of insertion site they are reliable. Complications include infections, catheter occlusions, vein occlusions, catheter disconnections, collections of fluid at the site, line leakage, vessel perforation, and device failure.
Brand name(s): CellCept
Why is this medication prescribed?
How should this medicine be used?
What special precautions should I follow?
What special dietary instructions should I follow?
What should I do if I forget a dose?
What side effects can this medication cause?
What storage conditions are needed for this medicine?
In case of emergency/overdose
What other information should I know?
IMPORTANT WARNING:
Mycophenolate makes you more susceptible to illnesses. Avoid people with contagious diseases such as the flu and colds. Keep cuts and scratches clean. Use good personal hygiene, especially for your mouth, teeth, skin, hair, and hands.
Why is this medication prescribed?
Mycophenolate is used to prevent rejection of heart or kidney transplants.This
medication is sometimes prescribed for other uses; ask your doctor or
pharmacist for more information.
How should this medicine be used?
Mycophenolate comes as a capsule, tablet, and suspension to take by mouth. It is usually taken two times a day. Follow the directions on your prescription label carefully, and ask your doctor or pharmacist to explain any part you do not understand. Take mycophenolate exactly as directed. Do not take more or less of it or take it more often than prescribed by your doctor.
Do not crush tablets or open or crush the capsules. Avoid inhaling the powder or allowing the powder to touch your skin. If you accidentally come in contact with the powder, use soap and water to wash the area.
What special precautions should I follow?
Before taking mycophenolate:
- tell your doctor and pharmacist if you are allergic to mycophenolate or any other drugs.
- tell your doctor and pharmacist what prescription and nonprescription medications you are taking, especially acyclovir (Zovirax), antacids, aspirin, azathioprine (Imuran), cholestyramine (Questran, Prevalite), colestipol (Colestid), ganciclovir (Cytovene), phenytoin (Dilantin), probenecid (Benemid), salicylates, theophylline (Theo-Dur), and vitamins. You should not take an antacid within 1 hour before or 2 hours after mycophenolate.
- tell your doctor if you have or have ever had kidney disease, skin cancer, stomach problems, or any other gastrointestinal disease.
- tell your doctor if you are pregnant, plan to become pregnant, or are breast-feeding. If you become pregnant while taking mycophenolate, call your doctor immediately. If you are of childbearing potential, you must use contraception at least 6 weeks before beginning mycophenolate and should continue contraception for 6 weeks after you stop taking this medicine. Two forms of birth control must be used during therapy unless abstinence is the chosen method.
- if you are having surgery, including dental surgery, tell the doctor or dentist that you are taking mycophenolate.
- you should plan to avoid unnecessary or prolonged exposure to sunlight and to wear protective clothing, sunglasses, and sunscreen. Mycophenolate may make your skin sensitive to sunlight.
What special dietary instructions should I follow?
Mycophenolate should be taken on an empty stomach. If there are problems with nausea, the medication can be taken with a few crackers to alleviate it.
What should I do if I forget a dose?
Take the missed dose as soon as you remember it. However, if it is almost time for the next dose, skip the missed dose and continue your regular dosing schedule. Do not take a double dose to make up for a missed one.
What side effects can this medication cause?
Although side effects from mycophenolate are not common, they can occur. Tell your doctor if any of these symptoms are severe or do not go away:
- diarrhea
- vomiting
- stomach pain
- headache
- tremor
- insomnia
- dizziness
If you experience any of the following symptoms, call your doctor immediately:
- bloody vomit
- bloody diarrhea or black, tarry stools
- fever
- unusual bruising or bleeding
What storage conditions are needed for this medicine?
Keep this medication in the container it came in, tightly closed, and out of reach of children. Store it at room temperature and away from excess heat and moisture (not in the bathroom). Throw away any medication that is outdated or no longer needed. Talk to your pharmacist about the proper disposal of your medication.
In case of overdose, call your local poison control center at 1-800-222-1222. If the victim has collapsed or is not breathing, call local emergency services at 911.
What other information should I know?
Keep all appointments with your doctor and the laboratory. Your doctor will order certain lab tests to check your response to mycophenolate. This will be a CBC/differential (complete blood count) and a trough (blood taken before the first dose of the day is given) Cellcept level.
Before receiving any vaccinations, tell your doctor that you are taking mycophenolate. NO FLU SHOTS or vaccinations in OMS children!!
Do not let anyone else take your medication. Ask your pharmacist any questions you have about refilling your prescription.